Zachary Huang, Michigan State University
Jointly published in the American Bee Journal and in Bee Culture, September 2011 and also at extension.org/bee_health
URL: https://bee-health.extension.org/effects-of-nosema-on-honey-bee-behavior-and-physiology/
Currently there are two species of Nosema infecting the Western honey bees, Apis mellfiera. Nosema apis is the first species described by Zander in 1909. In 1995, Ingmar Fries described a new species of Nosema in the Asian honey bee, Apis cerana, thus it was named Nosema ceranae. It was thought that N. ceranae mostly infected A. cerana, although there was a mention that A. mellifera could become infected with N. ceranae under laboratory conditions. In 2005, natural infection of N. ceranae was reported in A. mellifera colonies from Taiwan (Huang et al., 2005). Shortly thereafter, the infection of N. ceranae to A. mellifera was reported in Europe, United States, China, and worldwide. In this article I will review old studies done on N. apis and recent studies on N. ceranae, from a behavioral and physiological perspective. For basic biology, diagnosis and control of N. ceranae, please refer to Tom Webster’s article at here.
1. Differences among the two species
The differences between the two Nosema species are listed in Table 1, but I will only discuss differences in morphology and symptoms here, other differences are dealt with in more detail in the following sections. N. ceranae spores are slightly smaller, less symmetrical and the two ends are sharper, compared to N. apisspores (Fig. 1). Cross sections of N. ceranae spores show a fewer number of coils of polar filament compared to N. apis spores. N. ceranae is more resistant to high temperature, Fenoy et al (2009) claims that at 60°C for 1 month, over 90% of N. ceranae spores were still viable. N. cenarae spores lost ~90% infectivity at freezing temperature for one week, while N. apis spores retained 100% of its activity. The typical symptoms for N. apis infection are also lacking in N. ceranae infected bees: such as defecation near or inside the hive entrance during winter (Fig. 2), and milky color of midgut in heavily infected bees (Fig. 3). Spanish studies claim that N. ceranae infection rates do not show the typical changes with season, while prior studies of N. apis indicate that infection rate drops down in summer but stays high in spring.

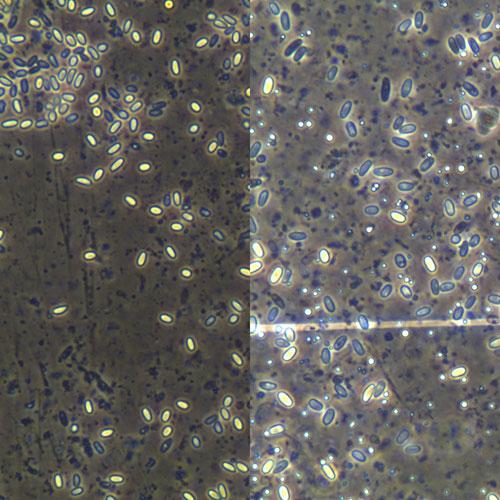
Fig. 1. Spores of Nosema ceranae (left) and Nosema apis (right) and under the same magnification (400x) using a compound microscope. N. ceranae is about 20% smaller and amore almond shaped.
2. Is N. ceranae more virulent than N. apis?
In a widely cited study conducted in Spain (Higes et al. 2007), bees infected with N. ceranae in the laboratory cages showed 94.1% and 100%mortality seven and eight days after inoculation, respectively. Although N. apis was not compared in the same experiment, by comparing cage studies using N. apis conducted earlier, it was suggested that N. ceranae is much more virulent than N. apis. More recent studies from other laboratories failed to see this difference. Forsgen and Fries (2010) compared the mortality of bees infected with either species and did not see a difference between the two. The MSU honey bee laboratory also failed to see a difference in mortality between bees infected with either species of Nosema (Z.Y. Huang, unpublished data). It is not clear whether the Spanish Nosema strain is more virulent, or whether the Spanish honey bees (Apis mellifera iberica) are more susceptible to N. ceranae.
Table 1. Differences between N. apis and N. ceranae.
| N. apis | N. ceranae | Section | |
| Spore size | 6 x 3 μm | 4.4 x 2.2 μm | 1 |
| Spore morphology | Ends rounded, symmetrical | Ends sharper,less symmetrical | |
| Polar filament coils | >30 | 18-21 | 1 |
| Resistance to high temperature | Low | High | 1 |
| Resistance to freezing | High | Low | 1 |
| Seasonal fluctuations | May have infections in summer | Lack of seasonality | 1 |
| Defecation near or inside the hive | Yes | No | 1 |
| Milky color of midgut | Yes | No | 1 |
| Virulence | Low | Higher | 2 |
| Earlier foraging | Yes | unknown | 3 |
| Enhanced JH production | Yes | unknown | 3 |
| Homing ability | Likely reduced | Reduced | 4 |
| Energy cost | Low | High | 4 |
| Immuno-supression | No | Yes | 4 |
3. Effects by Nosema apis
3.1. Effects on workers and queens
In the 1990s, T.P. Liu in Canada conducted many studies, most of them at the ultra-structural level on the effects of N. apis on honey bees. His studies indicated that workers infected with N. apis show ultrastructural changes in the cells from midgut epithelium, hypopharyngeal glands, and corpora allata (sources of juvenile hormone). Oöcytes in queens infected with N. apis for only 7 days were already degenerated. The ovariole sheath became wrinkled. In the oöplasm, yolk granules broke down into small spheres and granular substances and the oöcytes became extensively autolyzed. It is not clear whether the oocyte degeneration in infected queens is due to a pathological process, a lack of protein nutrition, or to increased juvenile hormone production as a result of Nosema infection (see below).


Fig. 2. Midgut (ventriculs) tissue of a bee infected by N. apis (top) and a healthy bee (bottom). Healthy bee midguts are straw colored, translucent and ring like structure can be seen, while infected midguts are milky and the structures are not as clear. It was said N. ceranae infection does not show this symptom, which is typical of N. apis.
3.2. N. apis causes earlier foraging, and higher JH production
Worker bees infected by N. apis have smaller hypopharyngeal glands and show an earlier regression in gland size than un-infected bees. In addition, Nosema infected bees show a more rapid behavioral maturation than un-infected bees (Wang and Moeller, 1970). Infected bees also guarded more frequently, and performed the following behaviors earlier than un-infected bees: orientation flight, dance following, and foraging. Infected bees also show a decreased tendency to feed the queen. As predicted by a theoretical model, which states that workers should take more risks when they are parasitized or “not as worthy to the society” as others, Woyciechowski and Kozlowski (1998) demonstrated that N. apis infected workers showed increased foraging activity than healthy foragers during adverse weather conditions.
The changes in N. apis infected bees are very similar to those induced by artificially applying juvenile hormone (JH), which is low in nurses but high in foragers. Huang et al. (2001) studied whether the earlier foraging in infected bees was due to an early raise of JH. They found that infected workers foraged at an earlier age and showed higher haemolymph juvenile hormone (JH) titers than control bees in preforaging bees. This suggests that N. apis infection induces workers to forage earlier via higher JH titers. The higher JH titers could be achieved by several alternative mechanisms: enhanced JH production by host corpora allata (CA), reduced JH degradation, or JH production by Nosema directly. The same study found that rates of in vivo JH biosynthesis as well as JH degradation were higher in Nosema infected bees than control bees. Workers with their source of JH (CA) removed, but fed Nosema had no detectable levels of juvenile hormone in hemolymph and these bees did not forage early. These results suggest that Nosema-infected workers forage at an earlier age than control bees due to higher JH titers, which arise through increased JH production, and despite of the increased JH degradation in infected bees. The data also suggested that Nosema apis does not produce JH directly.
Most likely the smaller hypopharyneal glands and earlier foraging in N. apis infected workers are due to the fact that Nosema infects the epithelial cells of the midgut, therefore greatly reducing the host’s ability to digest pollen, which is needed for the development of glands. Impaired protein metabolism are deduced by lower proteolytic activity of the mid-gut, lower amount of amino acids in hemolymph, lower levels of proteins in the fat bodies (reviewed by Kralj and Fuchs, 2010) and lower protein levels in hemolymph in infected bees (Z.Y. Huang and T. Zhou, unpublished data). Inadequate nutrition could fail to increase vitellogenin, which normally inhibits JH production. Therefore JH increases prematurely in these bees with a poor protein nutrition.
It is not clear whether N. ceranae causes the same changes in workers as N. apis or not: e.g. earlier foraging and higher JH production. However based on that fact that both species affect the midgut epithelial cells, N. ceranae will most likely cause the same early foraging and enhanced JH production in workers. This is now being studied at Marla Spivak’s laboratory.


Fig. 3. Defecation on hive bodies by bees with dysentery. This occurs in Midwestern U.S. in March to May. Dysentery can be caused by many factors, one of which is a N. apis infection. N. ceranae seems to cause this symptom occasionally, but it is not clear if it is due to mixed infection with N. apis.
4. Effects on bees
4.1 Learning and homing behavior affected by N. ceranae
When Kralj and Fuchs (2010) studied the homing behavior of bees mainly infected with N. ceranae, some bees were co-infected with N. apis. They found that infected bees released 6 and 10 m away from the colony took longer times to return. The percentage of bees that did not make home was higher in the infected bees compared to the healthy bees when released 30 m away from the colony. They also found a lower rate of infected bees among the returning foragers compared to departing foragers, suggesting some infected bees did not return home successfully. It is not clear why infected bees did not return home as well. The study used bees of known ages, so this is not because infected bees were developing precociously. The alternative is that infected bees did not have proper protein nutrition which affected their brain development and capacity of learning. It is not clear whether N. apis causes the same effect in honey bee learning and homing behavior. We have tried to determine if N. apis infected bees drifted more to surrounding colonies but failed to find if this is the case (Z.Y. Huang and H. Lin, unpublished data).
4.2. N. ceranae causes higher energy costs
Mayack and Naug (2010) compared the effect of energy stress on healthy and N. ceranae infected bees and found that N. ceranae infection caused an energy stress in bees from several lines of evidence. First, bees infected with N. ceranae were more responsive to sucrose solution (not as picky as healthy bees) and would extend their proboscis at a lower sugar concentration. Second, infected bees consumed about 87µl of 30% sucrose solution in 24 hours, while healthy bees consumed only 60µl. Therefore it appears that N. ceranae made the bees more “hungry” and caused them to drink more syrup. Third, control bees fed with 5, 10, 20 or 30 µl of syrup survived better compared to infected bees fed the same amounts during a 24 hour period. However if bees were fed ad libitum, or not fed at all, the two groups did not show any difference in their survival. The authors therefore concluded that N. ceranae caused an energy stress in infected bees, and speculated that this might be the main reason for the shortened survival of infected bees inside colonies.
Martín-Hernández et al. (2011) compared the energy cost of both Nosema species. Using caged bees, they showed that both mortality and sugar syrup consumption are the highest in N. ceranae infected bees, intermediate in N. apis infected bees and lowest in uninfected control bees. This study further demonstrates that N. ceranae has subtly different effects on honey bees compared to N. apis.
4.3. N. ceranae causes immune suppression
Antúnez et al. (2009) studied the immune response of honey bees after infection with either N. apis or N. ceranae. They measured gene expressions of several antibiotic peptides, abaecin, defensin and hymentoptaecin, produced inside honey bees after bacterial infection. In all three genes, N. apis infection caused an elevation of gene expression in either 4 or 7 days post infection, but N. ceranae did not show any difference in gene expression compared to the control (uninfected bees), or even significantly reduced it (abaecin at 7 days). These data suggest that N. ceranae actively suppresses the immune response in infected honey bees while N. apis does not.
Alaux et al. (2010) studied whether a neonicotinoid (imidacloprid) and Nosema (a mixture of both species) would show a synergistic interaction in affecting honey bees. They found that the combination of both agents caused the highest mortality and food consumption. They also found that the activity of glucose oxidase, an enzyme bees use to sterilize colony and brood food, was significantly decreased only by the combination of both factors compared with control, Nosema or imidacloprid only groups, suggesting a synergistic interaction between the two agents. Because the combined group showed similar Nosema spore counts to that of Nosema infected bees alone, it seems that the synergistic effect is due to the immune suppression of N. ceranae, causing bees to be more sensitive to the pesticide, rather than the pesticide reducing bee resistance to allow a more severe damage by Nosema.
In a more recent study, Vidau et al. (2011) found a similar synergistic effect between pesticides and N. ceranae. After being exposed to sublethal doses of fipronil or thiacloprid, N. ceranae-infected bees showed a higher mortality than in uninfected ones. The synergistic effect of N. ceranae and insecticide on honeybee mortality was not linked strongly to a decrease of the insect detoxification enzymes. This is because N. ceranae infection induced an increase in glutathione-S-transferase activity in midgut and fat body but not in the 7-ethoxycoumarin-O-deethylase activity. It is not clear how tightly the insect detoxification system and the immune system are linked – they might well not be tightly linked since one is induced by pesticides and another by parasites.
4.4. N. ceranae affects queen health
Alaux et al. (2011)studied the effect of N. ceranae infection on 8 day old honey bee queens. They found that N. ceranae did not affect the fat body content, which is an indicator of energy stores, but changed the vitellogenin titer, which is an indicator of fertility and longevity, the total antioxidant capacity and the queen mandibular pheromones. The strange thing is that these changes were contrary to the predicted direction that they were all increased in Nosema-infected queens. It is possible that these are only seen in 8 day old queens, perhaps due to accelerated development as seen in N. apis infected worker bees. It is not clear whether in older queens these changes will remain or become reversed.
5. Conclusions
Studies on N. apis were mostly done during the 1970-1990s, but there is a huge interest in N. ceranae since 2007, especially after Colony Collapse Disorder (CCD) appeared in this country. N. ceranae infection by itself does not seem to explain colony loss in the US, but in Spain it was speculated to be the main cause for CCD. There are clear differences in how the two species of Nosema affect our bees (Table 1). However, there are still many unanswered questions in the biology, epidemiology, and pathology of N. ceranae despite of the many studies. We are not even certain of how many of the old studies attributed to N. apis might be actually from N. ceranae, and how long N. ceranae has been in the U.S. Until now we did not have a single sample showing that prior to a certain time, we only had N. apis and N. ceranae came after that point. The notion that N. ceranae is replacing N. apis is also based on circumstantial evidence, however with ongoing monitoring of whether and how N. apis is slowly disappearing might help clarify this.
Acknowledgement
I thank Randy Oliver, Xianbing Xie, Julie Adams, Mike Jessup, and Alice Jiang for reviewing this manuscript. Research by the author’s laboratory on N. apis was supported by USDA NRI grant (97-35302-5304), and N. ceranae by Managed Pollinator CAP USDA NIFA 20098511805718.
Main references
- Alaux, C., J.L.Brunet, C. Dussaubat, F. Mondet, S. Tchamitchan S, et al. 2010. Interactions between Nosema microspores and a neonicotinoid weaken honeybees (Apis mellifera). Environ Microbiol 12: 774–782.
- Chen, Y, Huang, ZY. 2010. Nosema ceranae, a newly identified pathogen of Apis mellifera in the U.S. and Asia. Apidologie 41: 364-374.
- Higes, M, P. Garcia-Palencia, R. Martin-Hernandez, A. Meana. 2007. Experimental infection of Apis mellifera honeybees with Nosema ceranae(Microsporidia), J. Invertebr. Pathol. 94: 211–217.
- Kralj, J., S. Fuchs, 2010. Nosema sp. influences flight behavior of infected honey bee (Apis mellifera) foragers. Apidologie 41: 21–28.
- Liu, T.P. 1992, Oocytes degeneration in the queen honey bee after infection by Nosema apis. Tissue Cell 24: 131–138.
- Mayack, C., D. Naug, 2009 C. Mayack and D. Naug, Energetic stress in the honeybee Apis mellifera from Nosemaceranae infection, J. Invertebr. Pathol.100: 185–188
Originally posted 9/11/2011, updated 2/21/2021, fixed images, typos, and some words running together (copying/pasting sometimes causes this, not sure why).

Thank for the info I am always fascinated by honey bees and their functions.